Throwing away spoiled silage is no fun, it adds to feed costs and is labour intensive. Even worse, feeding poor quality silage has reduced nutritional value and contains undesirable microbes, along with their end-products can negatively affect herd health and productivity.
Even crops properly harvested and ensiled can be susceptible to undesirable fermentations and/or microbial activity. Silage can appear to ferment well and then heat and spoil during feedout. When good feed goes bad, it’s all down to the balance of microbes within the storage structure.
Ensiling forages correctly is a major opportunity to reduce undesirable spoilage microbes with a four-pronged approach:
- Start with a forage crop harvested at proper stage of maturity, moisture content and chop length
- Use the d a forage inoculant that adds efficient, research-proven lactic acid bacteria (LAB) according to your needs
- Pack the forage tight to exclude oxygen, which is the worst enemy of the ensiling process
- Cover and seal the structure to reduce further oxygen exposure
The goal of silage is to preserve the quality and nutrients present in those crops at the time of harvest, and it is important to start with high quality forage.
Dropping the pH (fast)
These recommendations are aimed to consume residual oxygen and stimulate fermentation to drop the pH of the forage mass. A low pH is needed to maintain stable, high-quality silage. It helps create the environment to essentially “pickle” the forage and coupled with lack of oxygen, helps prevent growth of spoilage microbes like clostridia, yeasts and molds. A delay in this step allows for extensive plant respiration and enzymatic activities, as well as allowing aerobic microorganisms to continue to degrade valuable nutrients, proliferate and generate heat (Table 1).
Table 1. Impact of delayed packing on the fungi population and levels of soluble sugars in fresh whole-plant corn
| 0h | 12h | 24h | |
|---|---|---|---|
| Yeasts (CFU/g) | 398,107 | 3,981,072 | 29,512,092 |
| Molds (CFU/g) | 301,995 | 6,025,596 | 69,183,097 |
| Sugars (% DM) | 3.7 | 1.9 | 0.6 |
Limiting amounts of soluble sugars coupled with low DM forages are perfect conditions for a clostridial fermentation. Certain species of clostridia can produce butyric acid and toxic biogenic amines, which can cause significant health and reproduction issues. Treating silage with a research-proven forage inoculant can help control clostridia growth. Moreover, formulations with fibrolytic enzymes help generate sugars necessary for a desired lactic fermentation.
Why the need for a silage inoculant?
Relying solely on the plants’ natural population of microorganisms for the fermentation process is a risky decision. At best, the result is normally a prolonged fermentation with a slow rate of pH drop with nutrients and DM losses.
Adding a silage inoculant is sometimes categorized as an insurance policy, but it is a lot more cost effective. For instance, a research-proven inoculant strain can maximize dry matter and nutrient retention, you can always make a good fermentation better! Plus, it can help improve aerobic stability to deliver stable, high quality and palatable feed. More significant challenges such as suboptimal harvest conditions may require an inoculant with a specific set of strains of LAB targeted to help control them.
Packing, packing, packing
Once the forage is delivered to the storage structure, oxygen must be excluded from the forage mass before the active fermentative phase takes place. Packing the forage tight is one of the most important and most overlooked factors when making good silage. Well-packed forage helps achieve an anaerobic environment, getting the fermentation started as quickly as possible, reducing DM losses. The minimum recommended packing density is 240 kg DM/m3 (44 pounds per cubic foot on a fresh weight basis or 15 pounds per cubic foot at 34 percent DM.)
Covering and sealing
It also is imperative to properly cover and seal the ensiling structure as soon as possible. If not properly covered, storage losses can be extensive due to air exposure and the effects of the elements. There has been reported losses of around 80% in the top 10 inches of different ensiled forage crops that were not covered. Additionally, the resulting “crust” is expected to have high numbers of detrimental microbes and potential metabolites that negatively impact animal intake, production and health.
Reducing oxygen exposure at feedout
As silage is opened and fed out, it is once again exposed to air. Oxygen allows lactate-assimilating, spoilage yeasts to “wake-up” and grow rapidly. These microbes use key nutrients in the silage, and this activity produces heat. Excessive heat can denature proteins and other nutrients in the silage. This is another opportunity for mold to grow on the silage and produce mycotoxins.
At this stage, common management practices can help reduce losses, including:
- Keeping the silage surface tight and clean
- Not removing silage too far ahead of feeding and leaving it sitting in loose piles, and/or
- Feeding out at the correct rate
- Use a silage defacer to remove silage
In most cases, aerobic instability and silage heating is caused by spoilage yeast growth, so all the management tips outlined above including using a proven inoculant to drive a fast, efficient fermentation will help. In addition, yeasts have been proven to reduce digestibility and feed intake (Table 2).
Table 2. Effect of spoilage yeasts on TMR stability and intake of dairy heifers
| Fresh | Spoiling | |
|---|---|---|
| Yeasts, CFU/g | 110,000 | 66,000,000 |
| pH | 4.16 | 5.17 |
| DMI, lb/d | 11.69 | 10.54 |
Watching for mold growth
In most cases, the yeasts that cause spoilage use residual sugars and lactic acid to grow. As a result, the pH rises as lactic acid is consumed. Then, molds and other opportunistic microbes (bacilli, listeria, enterobacteria, etc.) begin to appear. By the time you see mold growth, aerobic spoilage is well advanced and significant losses have already occurred.
Molds tend to grow in hot spots where there is air (oxygen) present. This is typically in poorly sealed surface layers; corners or shoulders of ensiled forages; or where pockets of air were trapped or packing was inadequate.
Mold growth can cause palatability issues and reduce animal performance. Plus, some molds can produce mycotoxins under certain circumstances. “Mycotoxin” is a general term used for secondary metabolites, which have been shown to have some associated toxicity issues and are produced quite naturally by some fungi (molds). Mycotoxin production is sometimes stimulated by certain stressors that occur while the plant is growing in the field. There are species such as Fusarium and Penicillium molds that are derived from the field and have the potential to create mycotoxins later.
However, mycotoxins may be produced during all phases of production, from the growing crop all the way through to livestock feeding. Different mold strains, like Aspergillus flavus, can cause mycotoxin production during storage.
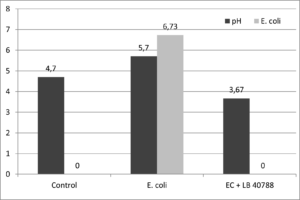
Unstable, high pH silages are susceptible to not only internal microbes but also external contamination. Cattle feeds, including silage, have been shown to transmit and permit the replication of E. coli and Salmonella species when contaminated in the manure. Researchers from the University of Florida studied corn silage re-inoculated with E. coli O157:H7 to simulate contamination with manure at feedout. Corn silage that had been inoculated with L. buchneri 40788 maintained a low pH and had undetectable counts of E. coli, while the pH of the untreated re-contaminated material increased, which allowed E. coli to grow (Figure 1)
Figure 1. The pH and E. coli counts (log CFU/g) of silages after 7 days of aerobic exposure.
Clean silage, tasting great
The benefits to hygienic clean silage management add up to more feed, better nutrition and increased palatability of the ration.
This document is based on the article by Renato Schmidt and John Brouillette and published in Progressive dairyman








